Abstract
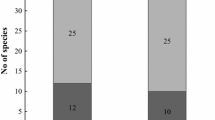
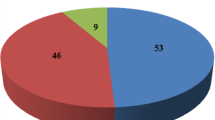
The present study aimed to assess the diversity of saprobic fungi associated with three decompositional stages of Hevea brasiliensis (Wild. ex Juss.) Mull. Arg. leaves along a chronosequence of plantations. The modified surface disinfection culture method was followed for isolation of saprobic fungi from three different phases (G1, G2 and G3) of decomposing leaves. The result revealed 51 species of fungi isolated from three plantations. There was an increasing trend of number of species along a chronosequence of plantations. Cladosporium sp. was isolated from all the phases of decomposition from all the three plantations. There is an increasing trend in diversity and a decreasing trend in dominance of fungi along a chronosequence of plantations. Overall, higher number of species and maximum diversity was recorded in G2 (Hʹ = 3.61) among the decompositional phases. The study suggests increase in diversity along a chronosequence of plantations and the similarity index suggests distinct fungal occurrence pattern in the leaf litter along a chronosequence of plantations.

Similar content being viewed by others
References
Allen SE, Grimshaw HM, Parkinson JA, Quaramby C (1974) Chemical analysis of ecological materials. Blackwell, Oxford
Anderson JM, Ingram JSI (1993) Tropical soil biology and fertility: a handbook of methods. CAB International, Wallingford
Anderson JM, Proctor J, Vallack HW (1983) Ecological studies in four contrasting lowland rain forests in Gunung Mulu National Park, Sarawak. J Ecol 71:503–527
Arunachalam A, Maithani K, Pandey NH, Tripathi RS (1998) Leaf litter decomposition and nutrient mineralization patterns in regrowing stands of a humid subtropical forest after tree cutting. For Ecol Manage 109:151–161
Bake TG, Attiwill PM (1985) Above-ground nutrient distribution and cycling in Pinus radiata D. Don and Eucalyptus obliqua D’Herit. forests in southeastern Australia. For Ecol Manage 13:41–52
Baldrian P, Lindahl B (2011) Decomposition in forest ecosystems: after decades of research still novel findings. Fungal Ecol 4:359–361
Berg B, Staaf H (1980) Decomposition rate and chemical changes in decomposing needle litter in Scots pine, in: Structure and function of northern coniferous forests. Ecolog Bulletins 32:373–390
Bills GF, Polishook JD (1994) Abundance and diversity of microfungi in leaf litter of a lowland rain forest in Costa Rica. Mycologia 86:187–198
Charley JL, Richards BN (1983) Nutrient allocation in plant communities, mineral cycling in terrestrial ecosystems. In: Lange OL, Nobel PS, Osmond CB, Ziegler H (eds) Physiological plant ecology IV. Ecosystem process. Mineral cycling, productivity and man’s influence. Springer, New York, pp 5–45
Chaudhuri PS, Bhattacharjee S, Dey A, Chattopadhyay S, Bhattacharya D (2013) Impact of age of rubber (Hevea brasiliensis) plantation on earthworm communities of West Tripura (India). J Environ Biol 34:59–65
De Boer W, Folman LB, Summerbell RC, Boddy L (2005) Living in a fungal world: impact of fungi on soil bacterial niche development. FEMS Microbiol Rev 29:795–811
Dickinson CH, Pugh GJF (1974) Biology of plant litter decomposition, vol. I and II. Academic Press, London
Domsch KH, Gams WH, Anderson TH (1980) Compendium of soil fungi, Vol-1. Academic Press, London
Ellis MB (1971) Dematiaceous hypomycetes. CAB International, Wallingford
Hirobe M, Sabang J, Bhatta BK, Takeda H (2004) Leaf-litter decomposition of 15 tree species in a lowland tropical rain forest in Sarawak: dynamics of carbon, nutrients, and organic constituents. J Fores Res 9:347–354
Kinkel LL, Andrews JH (1988) Disinfestation of living leaves by hydrogen peroxide. Trans Br Mycol Soc 91:523–528
Lodge DJ (1997) Factors related to diversity of decomposer fungi in tropical forests. Biodivers Conser 6:681–688
Lodge DJ, Cantrell SA, Gonzalez G (2014) Effects of canopy opening and debris deposition on fungal connectivity, phosphorus movement between litter cohorts and mass loss. For Ecol Manage 332:11–21
Magurran AE (2004) Measuring biological diversity. Blackwell Science Ltd., UK
McClaugherty CA, Berg B (1987) Cellulose, lignin and nitrogen concentrations as rate regulating factors in late stages of forest litter decomposition. Pedobiologia 30:101–112
Monkai J, Promputtha I, Kodsueb R, Chukeatirote E, McKenzie EHC, Hyde KD (2013) Fungi on decaying leaves of Magnolia liliifera and Cinnamomum iners show litter fungi to be hyper diverse. Mycosphere 4:292–301
Musvoto C, Campbell BM, Kirchmann H (2000) Decomposition and nutrient release from mango and miombo woodland litter in Zimbabwe. Soil Biol Biochem 32:1111–1119
Osono T (2005) Colonization and succession of fungi during decomposition of Swida controversa leaf litter. Mycologia 97:589–597
Osono T (2011) Diversity and functioning of fungi associated with leaf litter decomposition in Asian forests of different climatic regions. Fungal Ecol 4:375–385
Polishook JD, Bills GF, Lodge DJ (1996) Microfungi from decaying leaves of two rain forest trees in Puerto Rico. Microb Diversity 17:284–294
Prakash CP, Thirumalai E, Govinda RMB, Thirunavukkarasu N, Suryanarayanan TS (2015) Ecology and diversity of leaf litter fungi during early-stage decomposition in a seasonally dry tropical forest. Fungal Ecol 1(7):103–113
Puranong W, Lerstaveesin P, Ampornpa L, Sriktikulchai P (2007) Fungi associated to decomposition of Castanopsis accuminatissima leaf litter. In: 33rd Congress on Science and Technology of Thailand
Santana ME, Lodge DL, Lebow P (2005) Relationship of host recurrence in fungi to rates of tropical leaf decomposition. Pedobiologia 49:549–564
Schoening AG, Johansson G (1965) Absorptiometric determination of acid-soluble lignin in semi chemical bi sulfite pulps and in some woods and plants. Svensk Papperstid 68(18):607
Seephueak P, Petcharat P, Phongpaichit S (2010) Fungi associated with degrading leaf litter of para rubber (Hevea brasiliensis). Mycology 1:213–227
Seephueak P, Phongpaichit S, Hyde KD, Petcharat V (2011) Diversity of saprobic fungi on decaying branch litter of the rubber tree (Hevea brasiliensis). Mycosphere 2(4):307–330
Shanthi S, Vittal BPR (2010) Biodiversity of microfungi associated with litter of Pavetta indica. Mycosphere 1:23–37
Shanthi S, Vittal BPR (2012) Fungal diversity and the pattern of fungal colonization of Anacardium occidentale leaf litter. Mycology 3(2):132–146
Sharma G, Tharian GK, Dey SK (2014) Natural Rubber Sector in Tripura: Role of institutional Innovations. Agric Econom Res Rev 27:17–24
Sorensen T (1948) A method of establishing groups of equal amplitude in plant sociology based on similarity of species content. Det. Kong Danske Vidensk Selsk Biology Skr (Copenhagen) 5:1–34
Swift MJ, Russell-Smith A, Perfect TJ (1981) Decomposition and mineral-nutrient dynamics of plant litter in a regenerating bush-fallow in sub humid tropical Nigeria. J Ecol 69:981–995
Torreta NK, Takeda H (1999) Carbon and nitrogen dynamics of decomposing leaf litter in a tropical hill evergreen forest. Euro J Soil Biol 35:57–63
Updegraff DM (1969) Semi micro determination of cellulose in biological materials. Anal Biochem 3(2):420–424
Van Ryckegem G, Verbeken A (2005) Fungal ecology and succession on Phragmites australis in a brackish tidal marsh. I. Leaf sheaths. Fungal Divers 19:157–187
Vorıskova J, Petr Baldrian P (2013) Fungal community on decomposing leaf litter undergoes rapid successional changes. ISME J 7:477–486
Watanabe T (2002) Pictorial atlas of soil and seed fungi: morphologies of cultured fungi and key to species, 2nd edn. CRC Press LLC, NW Corporate Blvd, Boca Raton
Yamada A, Inoue T, Wiwatwitaya D, Ohkuma M, Kudo T, Abe T, Sugimoto A (2005) Carbon mineralization by termites in tropical forests, with emphasis on fungus combs. Ecol Res 20:453–460
Acknowledgements
The authors are thankful to Head, Department of Botany, Tripura University for providing the laboratory facilities. KC is grateful to Department of Science and Technology, Government of India for the INSPIRE fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chakraborty, K., Roy Das, A., Saha, A.K. et al. A culture based diversity of saprobic fungi associated with leaf litter of Hevea brasiliensis along a chronosequence of plantations in Tripura, Northeast India. Trop Ecol 61, 468–474 (2020). https://doi.org/10.1007/s42965-020-00104-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42965-020-00104-7